Stryker Rejuvenate and ABG II Modular Hhip Stem Litigation was recently coordinated in front of Judge Martinotti in the Supreme Court of New Jersey, Bergen
By: Indhira Benitez On Thursday, March 18th, at a hearing before a San Diego federal judicial panel on Multidistrict Litigation, Bayer’s lawyer, Marie Woodbury of
While the U.S. Food and Drug Administration (FDA) sets stringent requirements for approval of pharmaceuticals, it allows many medical devices to go to market without
Merck & Co. and subsidiary Schering-Plough are facing more lawsuits relating to yet another one of the company’s products, this time Vytorin. The manufacturers responsible
On January 17, 2013, the Food and Drug Administration issued a safety communication to health care providers, surgeons and patients who have received or are
By: George Mikhail The number of medical device products affected by recalls has increased the past eight quarters reaching over 123 million units recalled in
In the United States, an estimated 800,000 people rely on implantable defibrillators to regulate their heart rhythm. However, when this life-saving device starts malfunctioning, the
Invokana has now been associated with several amputations. As of May 2017, the FDA has sounded the alarm by adding warnings for this severe side effect.
Medical errors are the third leading cause of death in the United States. They are not the patient’s fault in any way, yet the Federal
German drugs and pesticides group Bayer said on Thursday it will pay around $1.6 billion to settle the majority of U.S. claims involving its Essure
The most common form of contraception, Intrauterine Devices (IUD), has been proven to cause dangerous outcomes in users. Medical studies have proven that unfavorable consequences
Intrauterine Devices (IUD) are a commonly utilized form of contraception used around the world by women. However, the most common brand, Paragard, has been linked
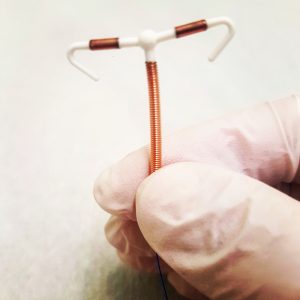
El DIU Paragard es uno de los DIU más populares del mercado y también es el centro de muchas demandas por parte de mujeres. Muchas
Paragard is a small IUD (intrauterine device) manufactured and marketed by Cooper Surgical. The contraceptive device is touted as 100%, safe, and efficient. It is
Paragard IUD is one of the most popular IUDs on the market – and it’s also the topic of many lawsuits by women. Many of

Biomet, the manufacturer of artificial hip M2a Magnum as well as a host of other orthopedic products faces a $21 million jury verdict after a

Since it was first approved by the FDA in 1984 and available to US consumers in 1988, the manufacturers of the contraceptive Paragard have boasted about its
By: George Mikhail With no surprise to our firm, the chair of the FDA hip implant panel, Dr. William Rohr of Mendocino Coast District Hospital,
By: George Mikhail Failing metal-on-metal hip replacements may be causing further medical complications by releasing toxic nano-particles. A new study published by the Journal of
By:George Mikhail Eight DePuy Orthopedics executives will be required by the court to testify in the ASR metal-on-metal hip implant litigation. DePuy Orthopedics, a subsidiary
By Paul Rheingold: A new article by New York City gynecologists using the Intuitive daVinci robot is the fullest explanation to date of the risks
By: George Mikhail Plaintiffs in lawsuits against Biomet are seeking to consolidate 40 nationwide lawsuits into a MDL (multidistrict litigation). Cases against Biomet are claiming
By: George Mikhail The Australian Therapeutic Goods Administration (TGA) has recognized the insurmountable evidence that points to failing metal-on-metal implants releasing chromium and cobalt in
By: Namra Ajmal Women are coming forward due to significant physical injury from Mirena; many women have experienced adverse side effects such as a dislocated
DePuy has been selling the Pinnacle hip implant longer than its ASR counterpart, although it has not been recalled. Nonetheless, adverse event reports are in
Our firm was the first to file NuvaRing suits in state and federal courts. Before any national or global attention was drawn to a higher
A new follow up study by Øjvind Lidegaard found that compared to combined oral contraceptives containing levonorgestrel users of vaginal ring contraceptives (such as NuvaRing)
A common, yet highly illegal, practice by large medical device manufacturers such as Medtronic is to offer financial kickbacks to doctors who use their products.
Medical devices can be an important part of patient care-but only if they work properly. According to patient safety advocates, more needs to be done
In April, the Archives of Surgery published a major article on the risks of robotic surgery by Buchs and others at the University of Illinois.
The Journal of Arthroplasty in May 2012, published a study titled “Metal-on-Metal Local Tissue Reaction Is Associated With Corrosion of the Head Taper Junction.” This
Experts in the United Kingdom are so alarmed with the failure rate of metal-on-metal hip implants that they have stated that the devices should no
When Bayer settled its Essure contraceptive lawsuits for $1.6 billion, the mega company resolved 90% of the nearly 39,000 defective product lawsuits pending against it.